clay science
It is estimated that all the clay on the planet, evenly spread out, would make a layer a mile thick - which is not surprising, given that clay is decomposed rock. What distinguishes clay from other types of "dirt" is particle size, as well as molecular and chemical properties. Size is very important: a grain of coarse sand falls through four inches of water in one second, a grain of silt takes five minutes, and a single grain of very fine clay can take up to 860 years! Clay is so slow partly because of shape as well as size - particles are thin and flat. Indeed, a single gram of clay can have a surface area larger than a football field. That flatness helps make clay plastic - flat surfaces make it easy for particles to stick, like two wet sheets of paper. In addition to size and shape, however, clay particles are also electrically charged, which means that they a tremendous capacity to attract and adhere to clay or other molecules (including water). All these properties make clay the wonderful, magical substance that it is.
RESOURCE William Bryant Logan assembled these facts in a marvellous book of essays titled, Dirt, The Ecstatic Skin of the Earth.
Mud, Mud, Glorious Mud
Suzanne Ubick
I often help my father-in-law make compost in the garden. Every time we finish a batch, he takes up a handful and says "I could eat this! Look at it, smell it, it just couldn't be any better!" Indeed, the compost is tempting: chocolate brown and crumbly, it looks like black forest cake and smells sweet and faintly earthy. Read more
A drawing of a menhir, or sacred stone,
at Ballymenach, Scotland.
It is marked by cupules where powder was ground from the rock for medicine.
IMAGE: CHARLES RAU
adsorption versus absorption
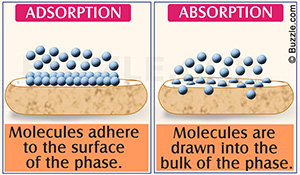
KEY UNDERSTANDING ABOUT CLAY'S POWER TO DETOXIFY:
"Adsorption" and "Absorption" : two words that look alike, but their difference is critical in understanding the functions of clay minerals. Read below to discover how positively charged toxins inside your cells, can be either bound or magnetised, by the negatively charged clay molecules and taken safely out of your body.
Adsorption characterizes the process by which substances stick to the outside surface of the adsorbent medium. The clay possesses unsatisfied ionic bonds around the edges of its mineral particles. It naturally seeks to satisfy those bonds. For this to happen, it must meet with a substance carrying an opposite electrical (ionic) charge. When this occurs, the ions held around the outside structural units of the adsorbent medium and the substance are exchanged.
The particles of clay are said to carry a negative electrical charge, whereas impurities, bacteria, or toxins, carry a positive electrical charge. For this very reason clay has been used to adsorb the colloidal impurities in beer, wine, and cider. The impurities in wine carry positive charges and can be agglomerated (brought together) and removed by stirring a small amount of negatively charged clay material into the wine. The clay particles attract the wine impurities and they settle out together (flocculate).
The process works the same in the human body. When clay is taken internally, the positively charged toxins are attracted by the negatively charged surfaces of the clay mineral. An exchange reaction occurs whereby the clay swaps its ions for those of the other substance. Now, electrically satisfied, it holds the toxin in suspension till the body can eliminate both.
The term active, or alive, indicated the ionic exchange capacities of a given clay mineral. The degree to which the clay-mineral ions become active determine its classification as alive. Living bodies are able to grow and change their form and size by taking within them lifeless material of certain kinds, and by transforming it into a part of themselves. No dead body can adsorb. It is physically impossible.
Absorption is a much more slow and involved process than adsorption. Here, the clay acts more like a sponge, drawing substances into its internal structure. In order for absorption to occur, the substance must undergo a chemical change to penetrate the medium's barrier. Once it has done that, it enters between the unit layers of the structure. Instead of the toxins, for instance, sticking only to the surface, they are actually pulled inside the clay. This is the reason why absorptive clays are labeled expandable clays. The more substances the clay absorbs into its internal structure, the more it expands and its layers swell.
Any clay mineral with an inner layer charge is an absorbent. Having an inner layer charge means having charged ions, sitting between layers, that are surrounded by water molecules. In this way, the clay will expand as the substance to be absorbed fills the spaces between the stacked silicate layers. Some clays are more gentle in their absorption, whereas others are definitely more radical. Absorption takes place with clay when the clay draws particulates into its internals layered structure, much like a sponge. Clay minerals have an inner layer charge that acts like an absorbent and can absorb and bond with many elements that are toxic, both man-made and natural.
Credit: shirleys-wellness-cafe.com
Clay as a toxin binder
From You Are The Healer : Dr. Sharol's
Aluminosilicate Clays And What To Expect From Them
Clay binders are used to assist removal of most polar types of toxins/biotoxins such as polar mycotoxins that individuals with "Chronic Inflammatory Response Syndrome (CIRS) due to water-damaged buildings", also called "Mold Related Illness" have trouble removing from their body.
People with mycotoxin/mold related illness are often unable to remove the mold toxins from their body due to improper antigen presentation or antigen acceptance by immune system cells/or other biotransformation/detox variants. The immune system of the CIRS individual or person with Mold Related Illness, is unable to remove these toxins and is the instigator of an inflammatory response. Aluminosilicate clays bind to some types of mycotoxins, or other biotoxins that cause CIRS allowing them to be eliminated via the feces. This only works for toxins that go through the enterohepatic circulation and end up in the bile. Some of all aluminosilicate clays may bind to bile acids, and help remove toxins attached to those bile acids, or they may bind directly to the toxin, but this is not known at this time. It is known that they do indeed bind and remove some mycotoxins in in vivo studies. The biotoxins must be removed from the body of the person with CIRS or they often to not get well. Once they are removed, the inflammatory process started by the biotoxins also needs to be addressed.
Zeolite Clinoptilolite: Therapeutic Virtues of an Ancient Mineral
Zeolites are porous minerals with high absorbency and ion-exchange capacity. Their molecular structure is a dense network of AlO4 and SiO4 that generates cavities where water and other polar molecules or ions are inserted/exchanged. Even though there are several synthetic or natural occurring species of zeolites, the most widespread and studied is the naturally occurring zeolite clinoptilolite (ZC). ZC is an excellent detoxifying, antioxidant and anti-inflammatory agent. As a result, it is been used in many industrial applications ranging from environmental remediation to oral applications/supplementation in vivo in humans as food supplements or medical devices. Moreover, the modification as micronization of ZC (M-ZC) or tribo mechanically activated zeolite clinoptilolite (TMAZ) or furthermore as double tribo mechanically activated zeolite clinoptilolite (PMA-ZC) allows improving its benefits in preclinical and clinical models. Despite its extensive use, many underlying action mechanisms of ZC in its natural or modified forms are still unclear, especially in humans. The main aim of this review is to shed light on the geochemical aspects and therapeutic potentials of ZC with a vision of endorsing further preclinical and clinical research on zeolites, in specific on the ZC and its modified forms as a potential agent for promoting human brain health and overall well-being.